Презентация Data Collection of Primary Central Nervous System (CNS) Tumors онлайн
На нашем сайте вы можете скачать и просмотреть онлайн доклад-презентацию на тему Data Collection of Primary Central Nervous System (CNS) Tumors абсолютно бесплатно. Урок-презентация на эту тему содержит всего 157 слайдов. Все материалы созданы в программе PowerPoint и имеют формат ppt или же pptx. Материалы и темы для презентаций взяты из открытых источников и загружены их авторами, за качество и достоверность информации в них администрация сайта не отвечает, все права принадлежат их создателям. Если вы нашли то, что искали, отблагодарите авторов - поделитесь ссылкой в социальных сетях, а наш сайт добавьте в закладки.
Презентации » Образование » Data Collection of Primary Central Nervous System (CNS) Tumors
Оцените!
Оцените презентацию от 1 до 5 баллов!
- Тип файла:ppt / pptx (powerpoint)
- Всего слайдов:157 слайдов
- Для класса:1,2,3,4,5,6,7,8,9,10,11
- Размер файла:1.89 MB
- Просмотров:66
- Скачиваний:0
- Автор:неизвестен
Слайды и текст к этой презентации:
№2 слайд


Содержание слайда: Portions of this presentation are based on non-malignant CNS tumor data collection rules adopted by the North American Association of Central Cancer Registries (NAACCR) Uniform Data Standards Committee - June 2003.
Portions of this presentation are based on non-malignant CNS tumor data collection rules adopted by the North American Association of Central Cancer Registries (NAACCR) Uniform Data Standards Committee - June 2003.
№4 слайд


Содержание слайда: Rationale for Non-malignant CNS Tumor Surveillance and Registration
Non-malignant CNS tumors cause disruption in normal function similar to that caused by malignant CNS tumors.
Location of a CNS tumor is as important as tumor behavior (benign or malignant) to morbidity and mortality.
№5 слайд


Содержание слайда: History 1992 -1996
1992 Central Brain Tumor Registry of the United States (CBTRUS) formed to report population-based data on primary benign, borderline, and malignant CNS tumors.
1996 National Coordinating Council on Cancer Surveillance (NCCCS) formed Brain Tumor Working Group (BTWG) to explore the feasibility of registering non-malignant CNS tumors
№7 слайд


Содержание слайда: BTWG Recommendations (1)
The following standard definition is to be used for collecting precise data for all primary intracranial and CNS tumors:
Primary intracranial and CNS tumors are all primary tumors occurring in the following sites, irrespective of histologic type or behavior:
№10 слайд


Содержание слайда: History 2000
International Classification of Diseases for Oncology 3rd Edition (ICD-O-3) and World Health Organization (WHO) 2000 Brain Tumor Classification are compatible.
November
Consensus conference on brain tumor definition convened. Group agrees to:
Site definition as in Recommendation 1.
Need to develop a standard site and histology definition based on the SEER site and histology validation list.
№11 слайд


Содержание слайда: History 2001-2002
2001 NCCCS
Accepted Recommendations 1 and 2 as completed.
Reconvened the BTWG to work on Recommendations 3 and 4.
2002 NAACCR established subcommittee of Registry Operations Committee to:
Identify changes needed in registry operations for inclusion of non-malignant CNS tumors.
October: Benign Brain Tumor Cancer Registries Amendment Act (Public Law 107-260) signed by President Bush.
№12 слайд


Содержание слайда: Reportable Brain-Related Tumors (1)
Public Law 107-260 requires reporting of brain-related tumors.
The term “brain-related tumor” means a listed primary tumor (whether malignant or benign) occurring in any of the following sites:
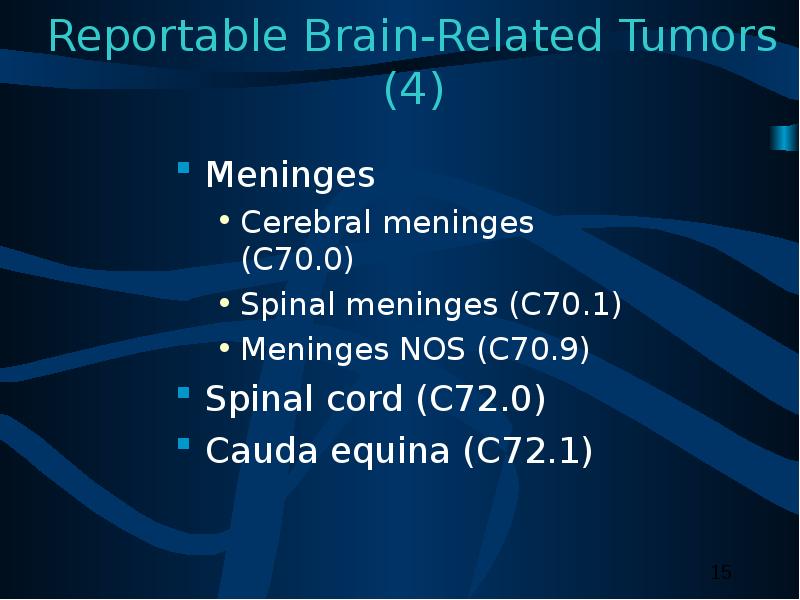
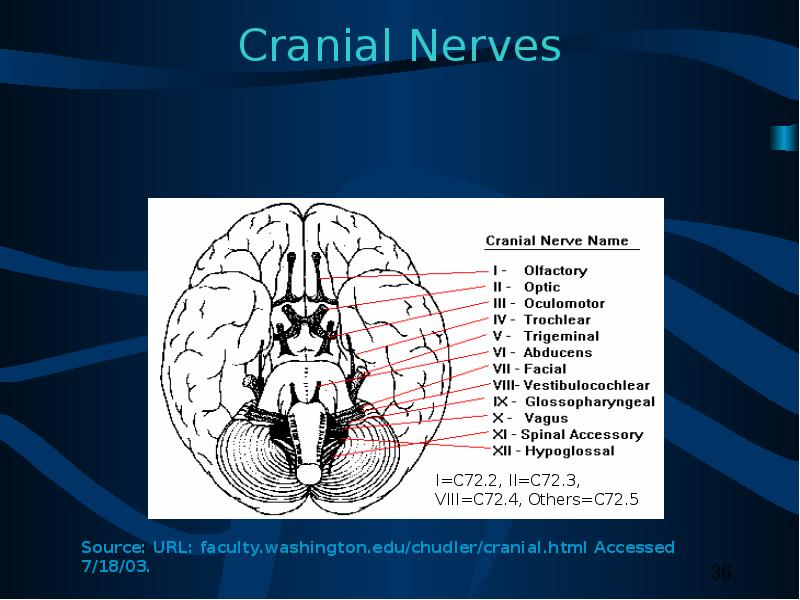
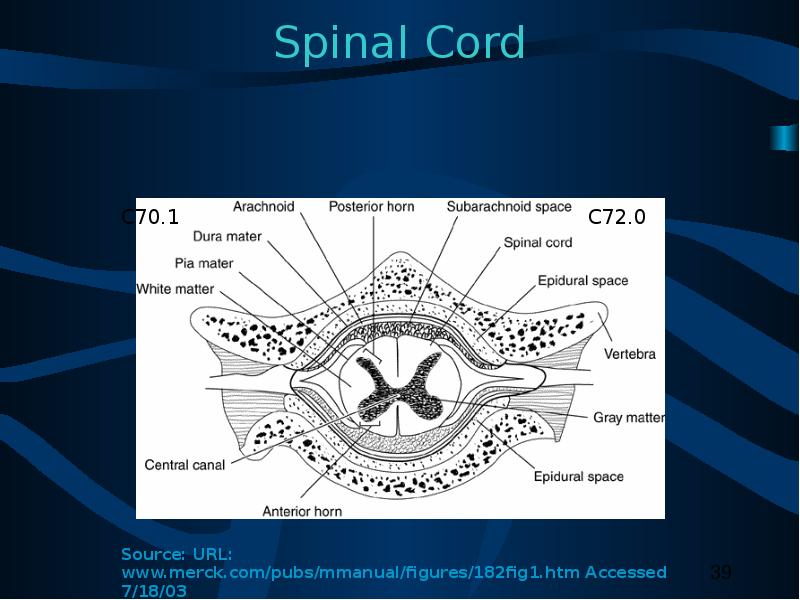
(I) The brain, meninges, spinal cord, cauda equina, a cranial nerve or nerves, or any other part of the CNS.
(II) The pituitary gland, pineal gland, or craniopharyngeal duct.
№19 слайд


Содержание слайда: Impact of Collecting Data on Non-malignant CNS Tumors (1)
Annual increase in number of cases estimated by doubling the number of malignant CNS cases diagnosed in the same year
Increase in hospital registry case load will depend on the type of hospital:
Community hospitals with small or no neurology service will likely experience a small increase in case load.
Hospitals with a large neurology service will likely experience a larger increase.
№20 слайд


Содержание слайда: Impact of Collecting Data on Non-malignant CNS Tumors (2)
Central registry case load is estimated to increase by 1%.
In 2002, 21 state cancer registries collected data on non-malignant CNS tumors:
Minimal impact if registry’s definition for brain-related sites does not change.
№27 слайд


Содержание слайда: Unusual and Ambiguous Terminology
If the final pathologic diagnosis is a CNS “neoplasm” or “mass”, an ICD-O-3 histology code must exist for the case to be reportable.
Hypodense mass or cystic neoplasm are not reportable, even for CNS sites.
A benign meningioma with a skull site should be coded to the cerebral meninges (C70.1).
№46 слайд


Содержание слайда: Non-Glial Tumors (2)
Meningiomas
Meningioma: Benign (M953_)
Malignant meningiomas
Anaplastic meningioma
Hemangiopericytoma (M9150)
Papillary meningioma (M9538)
Choroid plexus tumors
Choroid plexus papilloma (M9390)
Choroid plexus carcinoma
Choroid plexus meningioma (M9538)
№49 слайд


Содержание слайда: Other CNS Tumors (3)
Lymphomas (M9590)
Arise from
Indigenous brain histiocytes (microglia)
Rare lymphocytes in meninges
High incidence in patients with AIDS
Vascular tumors
Rare, non-malignant tumors
Arise from blood vessels of brain and spinal cord
Hemangioblastoma (M9161) most common vascular tumor
№50 слайд


Содержание слайда: Other CNS Tumors (4)
Cysts and tumor-like lesions
Reportable
Dermoid cysts (M9084)
Granular cell tumors (M9580)
Rathke pouch tumors (M9350)
Not reportable
Epidermoid cysts
Colloid cysts
Enterogenous cysts
Neuroglial cysts
Plasma cell granulomas
Nasal glial herterotopias
Rathke cleft cysts
№56 слайд


Содержание слайда: ICD-O-3 Coding Issues (1)
Some histologies may be difficult to determine if the primary site is intracranial or the skull (C41.0).
Non-malignant tumors of the skull are not reportable.
Chondroma (M9220/0) must originate in a brain-related site to be reportable.
Chordoma (M9370/3) and chondrosarcoma (M9220/3) are malignant.
Tumors in brain-related sites are analyzed separately from those in the skull.
№58 слайд


Содержание слайда: Grade for CNS Tumors
Sixth digit of ICD-O-3 histology code
Describes tumor differentiation or grade.
Is not usually specified for CNS tumors.
Is always assigned code 9 for non-malignant CNS tumors:
Not determined, not stated, or not applicable.
Per ICD-O-3, page 30, Rule G, paragraph 1 “Only malignant tumors are graded.”
Not the same as WHO grade.
№66 слайд


Содержание слайда: Determining Multiple Primaries:
Laterality
Brain is not a paired organ.
Laterality collected on both non-malignant and malignant tumors.
Used to determine if multiple non-malignant CNS tumors are counted as multiple primary tumors.
Not used to determine if multiple malignant tumors of the same intracranial or CNS site are multiple primary tumors.
№70 слайд


Содержание слайда: Determining Multiple Primaries
Malignant (1)
NO CHANGES (at this time)
Site
Rule: Each category (first three characters) as delineated in ICD-O-3 is considered to be a separate site.
Multiple tumors are:
Same: C71.0 Cerebrum, C71.2 Temporal lobe
Different: C70.0 Cerebral Meninges, C71.0 Cerebrum
№71 слайд


Содержание слайда: Determining Multiple Primaries:
Malignant (2)
Histology
Rule: Differences in histologic type refer to differences in the FIRST THREE digits of the morphology code.
Multiple tumors in the same site are:
Same: Choroid plexus carcinoma (M9390), Ependymoma (M9391)
Different: Astrocytoma (M9400), Gemistocytic astrocytoma (M9411)
№72 слайд


Содержание слайда: Determining Multiple Primaries
Non-malignant (1)
NEW RULES
Site
Rule: Each sub-site (fourth-digit level) as delineated in ICD-O-3 is considered a separate site.
Same site if separate tumors with the same histology are in the same sub-site.
Different site if separate tumors have the same histology in different sub-site
C71.1 Frontal lobe, C71.4 Occipital lobe
C70.0 Cerebral Meninges, C70.1 Spinal meninges.
№74 слайд


Содержание слайда: Determining Multiple Primaries
Non-malignant (3)
Site (cont)
Laterality: For non-malignant cases only
If multiple tumors of the same site and same histologic type are identified and both sides of a site listed as lateral are involved, tumors should be counted as separate primaries.
Different:
Right temporal lobe (C71.2) and left temporal lobe (C71.2)
№76 слайд


Содержание слайда: Determining Multiple Primaries:
Non-malignant (5)
Histology
If multiple tumors are in the same site, refer to Table 2, and use the following rules in priority order:
A-1: If the first three digits are the same but the codes are not found in Table 2, then the histology is considered to be the SAME.
A-2: If the first three digits are different but the codes are not found in Table 2, then the histology is considered to be DIFFERENT.
№77 слайд


Содержание слайда: Determining Multiple Primaries:
Non-malignant (6)
Histology (cont.)
B. If all histologies are listed in the same histologic group in Table 2, then the histology is considered to be the SAME. *
Example: Ependymomas: M9394, Myxopapillary ependymoma and M9444, Chordoid glioma have the same histology
*Note: If two histologies are in the same group in Table 2, code the first or more specific histology.
№78 слайд


Содержание слайда: Determining Multiple Primaries:
Non-malignant (7)
Histology (cont)
C: If the first three digits are the same as the first three digits for any histology in one of the groupings in Table 2 , then the histology is considered to be the SAME.*
Example: On table: Neuronal and neurol-glial neoplasm: M9505, ganglioglioma, Not on table: M9507, Pacinian tumor
* Note: If two histologies are in the same group in Table 2, code the first or more specific histology.
№79 слайд


Содержание слайда: Determining Multiple Primaries:
Non-malignant (8)
Histology (cont)
D: If the first three digits are the same and the histologies are from two different groups in the histologic groupings table, the histologies are considered to be DIFFERENT.
Example: Gliomas: M9442, Gliofibroma; Ependymoma: M9444, Chordoid glioma
№80 слайд


Содержание слайда: Determining Multiple Primaries:
Timing (1)
Primary malignant CNS tumors
NO CHANGE
Malignant tumors of the same site and same histology, diagnosed within 2 months:
Tumors are counted as the SAME primary.
Malignant tumors of the same site and same histology, diagnosed more than 2 months apart:
Tumors are counted as DIFFERENT primary sites.
№81 слайд


Содержание слайда: Determining Multiple Primaries:
Timing (2)
Primary non-malignant CNS tumors
NEW
No timing rule
If a new non-malignant tumor of the same histology as an earlier tumor that had been diagnosed in the same site is diagnosed subsequently at any time, it is considered to be the SAME primary tumor.
№83 слайд


Содержание слайда: General Rules for Determining Multiple Primaries of CNS Sites (2)
Multiple lesions: all non-malignant (cont.)
If same site and same histology:
Laterality is same side, one side unknown or not applicable, then SAME primary.
Laterality is both sides, then DIFFERENT primaries.
№84 слайд


Содержание слайда: General Rules for Determining Multiple Primaries of CNS Sites (3)
Multiple tumors: One non-malignant and one malignant
Non-malignant tumor followed by malignant tumor: DIFFERENT primaries, regardless of timing.
Malignant tumor followed by a non-malignant tumor: DIFFERENT primaries, regardless of timing.
№85 слайд


Содержание слайда: Histologic Transformation (1)
Histologic transformation or progression to a higher grade:
Determined by pathological review.
Final diagnosis made by review of previous biopsies or excisions and comparison to newly biopsied or resected brain tumor
Non-malignant tumor transforms to malignant tumor.
Malignant tumors transforms to higher grade tumor.
№87 слайд


Содержание слайда: Histologic Transformation (3)
Transformation of a non-malignant tumor to a malignant tumor is rare.
Malignant transformations include:
Changes from WHO grade I to WHO grade II, III, or IV.
Changes from behavior code 0 or 1 to code 2 or 3.
Complete two abstracts:
One for the non-malignant tumor
One for the malignant tumor
№88 слайд


Содержание слайда: Histologic Transformation (4)
Sequence Numbers
Non-malignant tumors: assigned sequence numbers from the reportable-by-agreement series.
Malignant tumors: assigned sequence numbers from the malignant series.
Example: Patient has a non-malignant CNS tumor that progressed into a malignant CNS tumor:
Non-malignant tumor is sequenced as 60.
Malignant tumor is sequenced as 00.
№89 слайд


Содержание слайда: Histologic Transformation (5)
Date of Diagnosis
Non-malignant tumors: First date that a medical practitioner diagnosed the non-malignant tumor either clinically or histologically.
Malignant tumors: First date that a medical practitioner diagnosed the malignant transformation either clinically or histologically.
№90 слайд


Содержание слайда: Coding Sequence Numbers (1)
Indicates the sequence of all reportable neoplasms over the lifetime of the person.
Codes 00 – 35: Malignant and in situ reportable neoplasms.
Codes 60 – 87: Reportable-by-agreement including non-malignant tumors diagnosed after January1, 2004.
№92 слайд


Содержание слайда: Coding Sequence Numbers (3)
Sequence numbers for non-malignant CNS tumors are assigned over the lifetime of the person.
Example: Patient diagnosed with a non-malignant CNS tumor in January, 2003 (not reportable by state or hospital reporting rules) and diagnosed with second non-malignant CNS tumor in 2004:
Second is sequence number 62.
Complete abstract for the second tumor only.
№97 слайд


Содержание слайда: Coding Collaborative Stage (2)
Site-specific codes for lymph nodes
Same for the Brain, cerebral meninges and other CNS.
Code 88: Not applicable.
For pituitary gland, craniopharyngeal duct, and pineal gland
Code 99: Not applicable.
Metastasis at Diagnosis
Same for the pituitary gland, craniopharyngeal duct, and pineal gland and other CNS.
Different for brain and cerebral meninges.
№98 слайд


Содержание слайда: CS Extension: Brain and Meninges
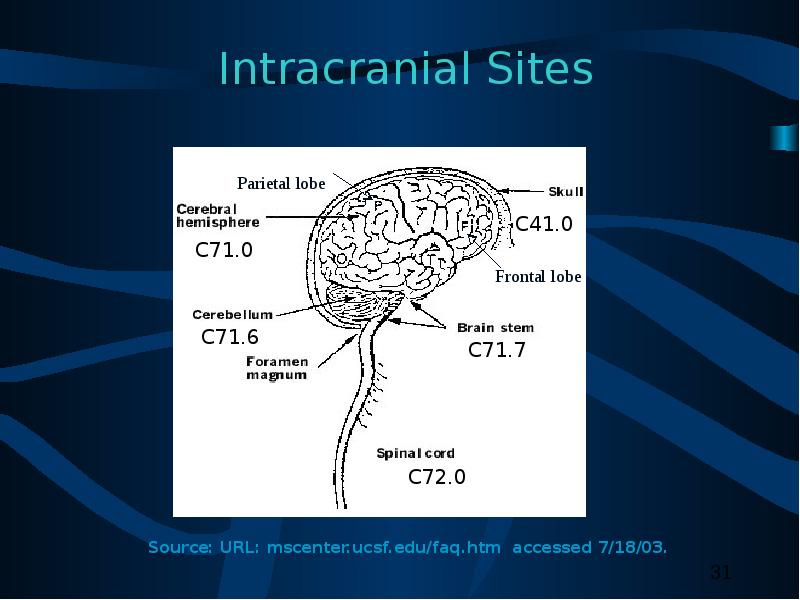
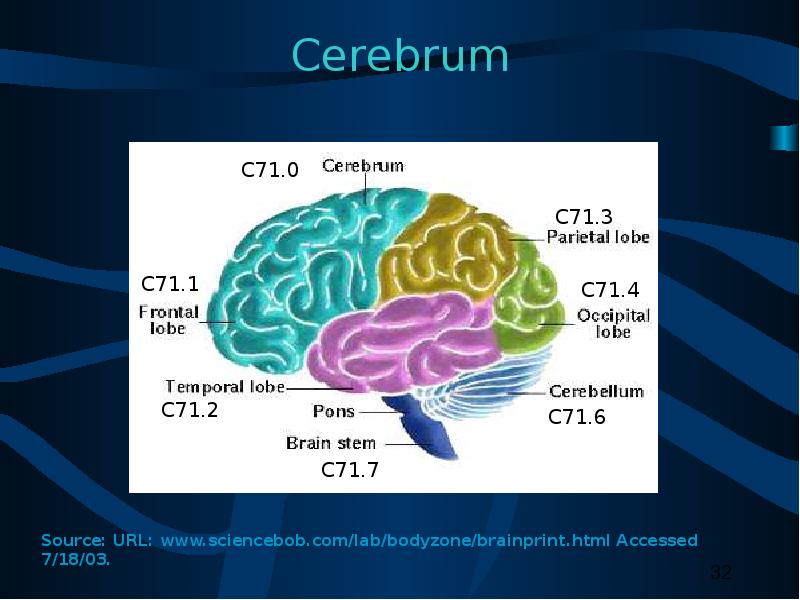
C70.0, C71.0 – C71.9 (1)
05 Benign or borderline brain tumors
10 Supratentorial tumor confined to CEREBRAL HEMISPHERE (cerebrum) or MENINGES of cerebral hemisphere one side: frontal lobe, occipital lobe, parietal lobe, or temporal lobe
11 Infratentorial tumor confined to CEREBELLUM or MENINGES of CEREBELLUM on one side: Vermis, lateral lobes, median lobe of cerebellum
№99 слайд


Содержание слайда: CS Extension: Brain and Meninges
C70.0, C71.0 – C71.9 (2)
12 Infratentorial tumor confined to BRAIN STEM or MENINGES of BRAIN STEM on one side: medulla oblongata, midbrain (mesencephalon), pons, hypothalamus, or thalamus
15 Confined to brain, NOS, Confined to meninges, NOS
20 Infratentorial tumor: Both cerebellum and brain stem involved with tumor on one side
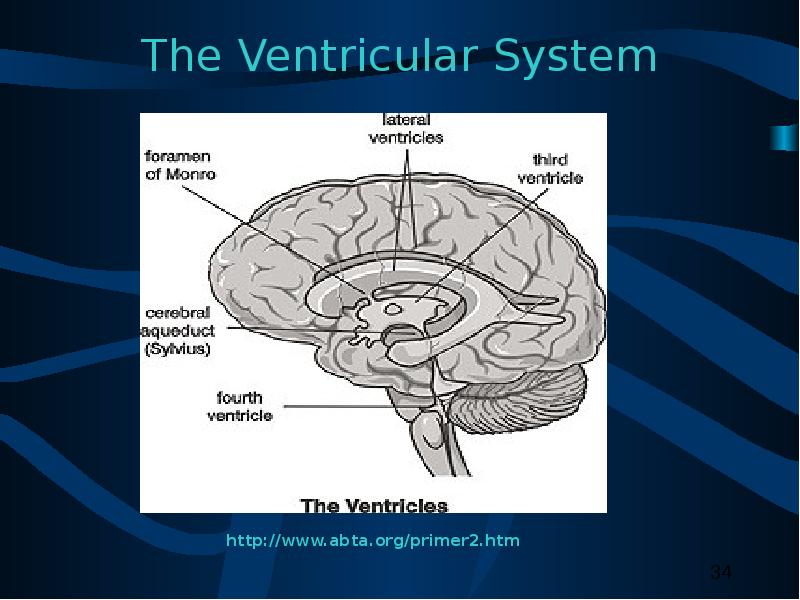
30 Confined to ventricles - Tumor invades or encroaches upon ventricular system
№100 слайд


Содержание слайда: CS Extension: Brain and Meninges
C70.0, C71.0 – C71.9 (3)
40 Tumor crosses the midline: involves the contralateral hemisphere, involves corpus callosum (including splenium)
50 Supratentorial tumor extends infratentorially to involve cerebellum or brain stem
51 Infratentorial tumor extends supratentorially to involve cerebrum (cerebral hemisphere)
60 Tumor invades bone (skull), major blood vessel(s), meninges (dura), nerves, NOS (cranial nerves), or spinal cord/canal
№101 слайд


Содержание слайда: CS Extension: Brain and Meninges
C70.0, C71.0 – C71.9 (4)
70 Circulating cells in cerebral spinal fluid; nasal cavity; nasopharynx; posterior pharynx; or outside CNS
80 Further contiguous extension
95 No evidence of primary tumor
99 Unknown extension; Primary tumor cannot be accessed; Not documented in patient record
№103 слайд


Содержание слайда: CS Extension: Other CNS
C70.1-9, C72.0–C72.9 (2)
05 Benign or borderline tumors
10 Tumor confined to tissue or site of origin
30 Localized, NOS
40 Meningeal tumor infiltrates nerve; nerve tumor infiltrates meninges (dura)
50 Adjacent connective/soft tissue; adjacent muscle
60 Brain, for cranial nerve tumors; major blood vessel(s); sphenoid and frontal sinuses (skull)
№104 слайд


Содержание слайда: CS Extension: Other CNS
C70.1-9, C72.0–C72.9 (3)
70 Brain except for cranial nerve tumors; bone, other than skull; eye
80 Further contiguous extension
95 No evidence of primary tumor
99 Unknown extension; primary tumor cannot be assessed; not documented in patient record
№105 слайд


Содержание слайда: CS Extension: Other Endocrine
C75.1, C75.2, C75.3
00 In situ; non-invasive; intraepithelial
05 Benign or borderline tumors
10 Invasive carcinoma confined to gland of origin
30 Localized, NOS
40 Adjacent connective tissue
60 Pituitary and craniopharyngeal duct: Cavernous sinus; infundibulum; pons; sphenoid body and siunses
Pineal: Infratentorial and central brain
80 Further contiguous extension
95 No evidence of primary tumor
99 Unknown extension
№106 слайд


Содержание слайда: CS Lymph Nodes
Describes tumor involvement of regional lymph nodes.
Code for CS Lymph Nodes is 88 (not applicable) for meninges, brain, spinal cord, cranial nerves, and other parts of the CNS.
Code for CS Lymph Nodes is 99 (unknown, not stated) for pituitary gland, craniopharyngeal duct, and pineal gland.
№108 слайд


Содержание слайда: CS Metastasis at Diagnosis
Other CNS and Other Endocrine
C70.1-9, C72.0—9, C75.1, C75,2, C75.3
00 No; None
10 Distant lymph node(s)
40 Distant metastasis except lymph nodes (code 10)
Distant metastasis, NOS
Carcinomatosis
50 (40) + (10)
99 Unknown; distant metastasis cannot be assessed; not documented in patient record
№113 слайд


Содержание слайда: Genetic Syndromes
Genetic syndromes associated with multiple CNS tumors are:
Neurofibromatosis I (von Recklinghausen’s disease)
Neurofibromatosis II (bilateral acoustic neurofibromatosis)
Von Hippel-Lindau disease
Tuberous sclerosis (Bourneville-Pringle syndrome)
Gorlin syndrome (Nevoid Basal Cell Carcinoma syndrome
Hermans-Grosfeld-Spaas-Valk disease
Li-Fraumeni syndrome
Familial retinoblastoma
Turcot syndrome (Adenomatous Polyposis syndrome)
Cowden disease
№129 слайд


Содержание слайда: Surgical Procedure of Primary Site
C75.1, C75.2, C75.3 (4)
Code 40: Total surgical removal of primary site; enucleation
Code 50: Surgery stated to be “debulking”
Code 60: Radical surgery
Partial or total removal of the primary site WITH resection in continuity (partial or total removal) with other organs
Code 90: Surgery, NOS
№131 слайд


Содержание слайда: Scope of Regional Lymph Node Surgery
Identifies removal, biopsy, or aspiration of regional lymph node(s):
NPCR-, COC-, and SEER-required data item.
Code 9: Meninges, brain, and spinal cord; cranial nerves; and other parts of the CNS.
Code as appropriate: Pituitary gland, craniopharyngeal duct, and pineal gland.
№132 слайд


Содержание слайда: Radiation Therapy (1)
Radiation codes indicate type of radiation therapy performed as part of the first course of treatment.
Records modality of radiation therapy used to deliver significant regional dose to the primary volume of interest.
COC-required data item.
SEER collects these data from COC-approved facilities
NPCR: Supplementary or recommended.
№133 слайд


Содержание слайда: Radiation Therapy (2)
Beam radiation
Codes 20 – 29:
Conventional radiation therapy: from an external beam directed at the tumor.
Energy is orthovoltage, cobalt, photon, and/or electron.
Code 30: Boron neutron capture therapy (BNCT)
Code 31: Intensity-modulated radiation therapy (IMRT)
№136 слайд


Содержание слайда: Radiation Therapy (4)
Radioactive implants
Code 50: Brachytherapy, radiation implants, radiation seeding, radioactive implants, interstitial implants, intracavitary radiation NOS
Code 51: Intracavitary radiation with low dose rate applicators (Cesium- 137, Fletcher applicator)
№137 слайд


Содержание слайда: Radiation Therapy (5)
Radioactive implants (continued)
Code 52: Intracavitary radiation with high dose rate applicator
Code 53: Interstitial radiation with low dose rate sources
Code 54: Interstitial radiation with high dose rate sources
Code 55: Low dose rate interstitial or intracavitary radium
№139 слайд

Содержание слайда: Chemotherapy (2)
Blood-brain barrier
Protects the brain from foreign substances, including chemotherapy.
May be disrupted by receptor-mediated permeabilizers.
Intrathecal chemotherapy
Drugs directly injected into the cerebrospinal fluid by spinal injection or Ommaya reservoir.
№142 слайд


Содержание слайда: Immunotherapy (1)
Record whether immunotherapeutic agents were administered as first course of treatment:
Angiogenesis inhibitors block the development of new blood vessels and starve the tumor.
Interleukins are growth factors that manipulate the tumor’s ability to grow.
№144 слайд


Содержание слайда: Hematologic Transplant and Endocrine Procedures
Identify systemic therapeutic procedures administered as first course of treatment:
Code 10: Bone marrow transplant, NOS
Code 11: Autologous bone marrow transplant
Code 12: Allogeneic bone marrow transplant
Code 20: Stem cell harvest
Code 30: Endocrine surgery and/or endocrine radiation therapy
Code 40: Combination of endocrine surgery and/or radiation with transplant procedure
№146 слайд


Содержание слайда: Technical Issues
Data Analysis Recommendations
Report and analyze data for non-malignant CNS tumors separately from malignant tumors.
Footnote that pilocytic astrocytomas are included in the analysis for malignant brain tumors for continuity of trends.
Review the standard site and histology groupings for tabulating estimates of these tumors to allow comparability of information across registries.
№147 слайд


Содержание слайда: References
Manuals, Articles, Reports
A Primer of Brain Tumors, 1998; American Brain Tumor Association, Des Plaines, IL; 800-886-2282 (can link to the manual through their website: www.abta.org)
Gershman S, Surawicz T, McLaughlin V, Rousseau V. Completeness of Reporting of Brain and Other Central Nervous System Neoplasms. Journal of Registry Management, Winter 2001, Volume 28, Number 4.
№148 слайд


Содержание слайда: References
Manuals, Articles, Reports (continued)
Fritz A, Percy C, Jack V, Shanmugaratnam K, Sobin V, Parkin D M , Whelan S. International Classification of Diseases for Oncology, 3rd ed. Geneva: World Health Organization, 2000
Report: Surveillance of Primary Intracranial and Central Nervous System Tumors: Recommendations from the Brain Tumor Working Group, National Coordinating Council for Cancer Surveillance, September 1998
№149 слайд


Содержание слайда: References
Websites
American Brain Tumor Association www.abta.org
American College of Surgeons, Commission on Cancer Information, Facility Oncology Data Standards (FORDS) www.facs.org/dept/cancer/index.html
American Joint Committee on Cancer, Collaborative Stage Documentation www.edits.cx/cs/
№150 слайд


Содержание слайда: References
Websites (continued)
Brain and Neurosurgery Information Center www.brain-surgery.com/index.html
Brain and Spinal Cord Tumors: Hope through Research www.ninds.nih.gov/health_and_medical/pubs/brain_tumor_hope_through_research.htm
Brain Tumor Guide http://virtualtrials.com/faq/toc.cfm
Central Brain Tumor Registry of the United States www.cbtrus.org/page2t.htm
№151 слайд


Содержание слайда: References
Websites (continued)
College of American Pathologists (CAP), Protocol – Brain ftp://ftp.cap.org/cancerprotocols/Brain03_p.doc
Illustrated Glossary of Radiology: Anatomy, Examinations and Procedures; Department of Radiology and Radiological Services, The Uniformed Services University of the Health Sciences
http://rad.usuhs.mil/glossary.html
№152 слайд


Содержание слайда: References
Websites (continued)
International RadioSurgery Association www.isra.org/index.html
National Brain Tumor Radiosurgery Association www.braintumors.com/radiosurgery/radiosrugery.info#TWO
NCI Brain Tumor Home Page www.nci.nih.gov/cancer_information/cancer_type/brain_tumor/
№153 слайд


Содержание слайда: References
Websites (continued)
PDQ Cancer Information Summaries: Adult Treatment www.cancer.gov/cancerinfo/pdq/adulttreatment
PDQ Cancer Information Summaries: Pediatric Treatment www.cancer.gov/cancerinfo/pdq/pediatrictreatment
The Brain Tumor Foundation www.braintumorfoundation.org/neurosurgery/ss3_3.htm
№154 слайд


Содержание слайда: Acknowledgments (1)
Prepared by
Shannon Vann, CTR
for the
North American Association of Central Cancer Registries (NAACCR)
This training presentation was supported by contract #200-2001-00044 from CDC. The content of this training presentation does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
№155 слайд


Содержание слайда: Acknowledgments (2)
Sponsors
Centers for Disease Control and Prevention
National Program for Cancer Registries
National Cancer Institute
Surveillance, Epidemiology and End Results Program
North American Association of Central Cancer Registries
American Joint Committee on Cancer
Collaborative Stage Task Force
№157 слайд


Содержание слайда: Acknowledgments (4)
Materials Review Committee
Trista Aarnes-Leong St. Vincent Medical Center, NAACCR Registry Operations Subcommittee,
Susan Bolick-Aldrich South Carolina Central Cancer Registry, NAACCR Registry Operations Subcommittee, Chair, Co-chair, Registry Operations Committee
Gayle Clutter CDC National Program of Cancer Registries, Registry Operations Subcommittee, National Coordination Council on Cancer Surveillance Brain Tumor Working Group, Chair
Faye Floyd CDC National Program of Cancer Registries
April Fritz NCI Surveillance, Epidemiology and End Results Program, Registry Operations Subcommittee
Elaine Hamlyn Canadian Cancer Registry, Registry Operations Subcommittee,
Holly Howe North American Association of Central Cancer Registries, Executive Director
Betsy Kohler New Jersey State Cancer Registry, NAACCR Education Committee
Carol Kruchko Central Brain Tumor Registry of the United States, Registry Operations Subcommittee, National Coordination Council on Cancer Surveillance Brain Tumor Working Group
Donna Morrel Cancer Surveillance Program of Los Angeles. Registry Operations Subcommittee
Linda Mulvihill North Carolina Central Cancer Registry, Registry Operations Subcommittee
Wendy Scharber Minnesota Cancer Surveillance Program
James Smirniotopoulos Professor of Radiology, Uniformed Services University, Registry Operations Subcommittee
Katheryne Vance California Cancer Registry, Registry Operations Subcommittee
Valerie Vesich American College of Surgeons, Commission on Cancer, Registry Operations Subcommittee
Скачать все slide презентации Data Collection of Primary Central Nervous System (CNS) Tumors одним архивом:
Похожие презентации
-
 Central Nervous System
Central Nervous System -
 Autonomic Nervous System
Autonomic Nervous System -
 Nervous system
Nervous system -
 Тестирование биржевых систем в условиях высокочастотного трейдинга Iosif Itkin Exactpro Systems LLC. - презентация
Тестирование биржевых систем в условиях высокочастотного трейдинга Iosif Itkin Exactpro Systems LLC. - презентация -
 Тестирование биржевых систем в условиях высокочастотного трейдинга Iosif Itkin Exactpro Systems LLC
Тестирование биржевых систем в условиях высокочастотного трейдинга Iosif Itkin Exactpro Systems LLC -
 Web 2. 0 is all about building systems that get better the more people use them Tim O'Reilly, 2005. - презентация
Web 2. 0 is all about building systems that get better the more people use them Tim O'Reilly, 2005. - презентация -
 PresentationID 1 2009 Cisco Systems, Inc. All rights reserved. Cisco Public. - презентация
PresentationID 1 2009 Cisco Systems, Inc. All rights reserved. Cisco Public. - презентация -
 Скрытый капитал открытых данных Кто и как может им воспользоваться в Беларуси? Презентация: Алексей Медвецкий, http://opendata. by.
Скрытый капитал открытых данных Кто и как может им воспользоваться в Беларуси? Презентация: Алексей Медвецкий, http://opendata. by. -
 Разработка, тестирование и развертывание баз данных в Visual Studio Team System 2010 Дмитрий Андреев dmitryanmicrosoft. com
Разработка, тестирование и развертывание баз данных в Visual Studio Team System 2010 Дмитрий Андреев dmitryanmicrosoft. com -
 Особенности использования TimesTen In-Memory Database в высоконагруженной среде Михаил Гранкин, QIWI
Особенности использования TimesTen In-Memory Database в высоконагруженной среде Михаил Гранкин, QIWI